Recent research has unveiled groundbreaking insights into protein behavior within cell membranes, revealing dynamic interactions that could revolutionize our understanding of cellular processes. On October 7, 2025, scientists reported these findings, highlighting the potential for transformative impacts on cellular signaling and function. Concurrently, studies have captured the destructive mechanism of Parkinson’s protein drilling holes in brain cells, offering a new perspective on neurodegenerative damage. This observation builds on earlier research from September 4, 2025, where scientists watched this protein action in real time.
Emerging Patterns in Membrane Protein Dynamics
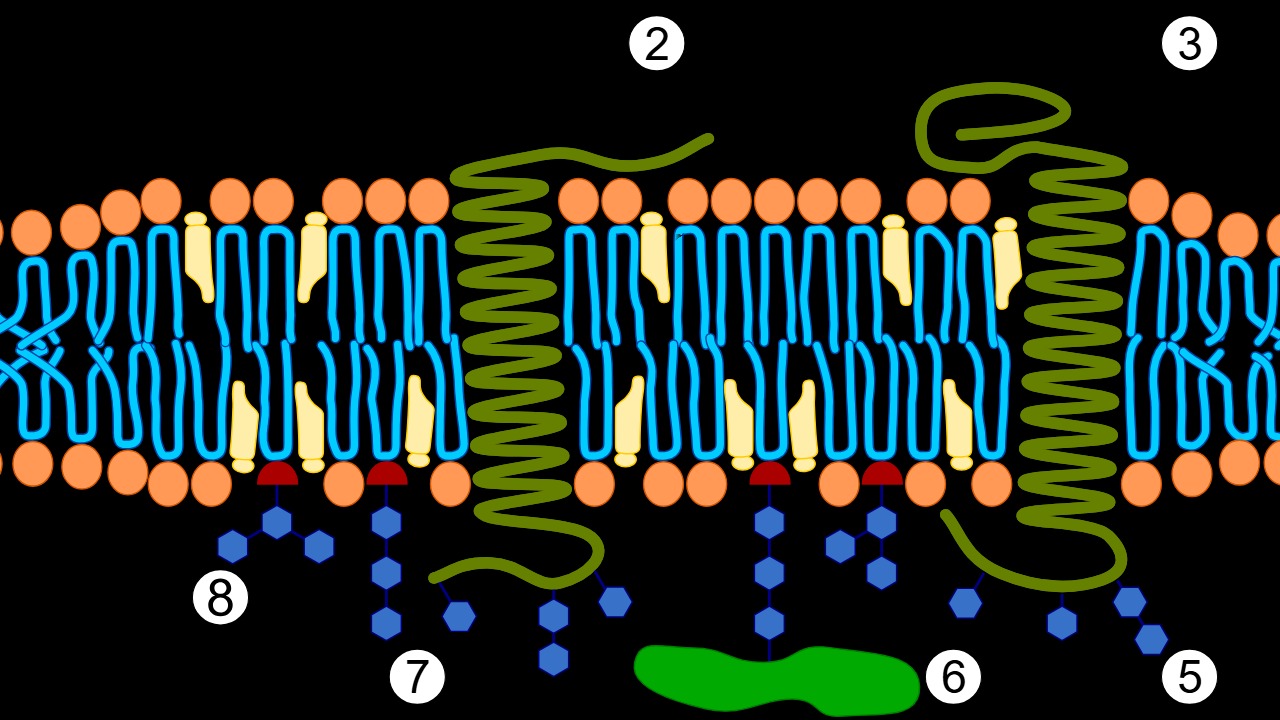
Scientists have recently unlocked new patterns of protein behavior in cell membranes, a discovery that could significantly influence our understanding of cellular signaling and function. These patterns, as reported on October 7, 2025, reveal structural and behavioral shifts in proteins embedded within cell membranes, suggesting a complex interplay that governs cellular processes (Phys.org).
The research highlights how these proteins, through their dynamic interactions, play a crucial role in cellular communication and response mechanisms. Advanced imaging techniques and simulations have been pivotal in observing these new patterns, providing a level of precision that underscores the significance of the discovery. This breakthrough could pave the way for novel therapeutic strategies targeting cellular dysfunctions.
These newly discovered patterns not only enhance our understanding of protein dynamics but also suggest potential applications in drug development. By targeting specific protein interactions within the membrane, researchers hope to design more effective treatments for diseases caused by cellular signaling errors. The study also emphasizes the role of lipid environments in modulating protein behavior, indicating that variations in lipid composition can significantly alter protein function. This insight opens up new avenues for exploring how changes in membrane composition might influence disease progression and treatment outcomes.
Furthermore, the research underscores the importance of interdisciplinary approaches, combining biophysics, molecular biology, and computational modeling to unravel the complexities of membrane protein dynamics. Such collaborative efforts are crucial for advancing our knowledge and developing innovative therapeutic strategies. The potential to manipulate these protein interactions could lead to breakthroughs in treating conditions like cancer and metabolic disorders, where cellular signaling plays a pivotal role.
Parkinson’s Protein Mechanism in Brain Cells
On October 7, 2025, scientists observed a Parkinson’s protein drilling holes in brain cells, a process that underscores the protein’s role in neurodegenerative damage. This finding provides critical insights into how such proteins contribute to cellular deterioration, potentially leading to conditions like Parkinson’s disease (SciTechDaily).
Previously, on September 4, 2025, researchers captured real-time footage of this protein drilling holes in brain cells, offering a detailed view of the process and its impact on cellular integrity. The visualization of this destructive mechanism highlights the protein’s ability to compromise brain cell function, linking it to broader patterns of protein behavior in membranes (ScienceDaily).
This destructive mechanism of Parkinson’s protein not only highlights its role in disease progression but also suggests potential targets for therapeutic intervention. By understanding how these proteins create structural damage in brain cells, researchers can develop strategies to inhibit their activity or prevent their formation. This could lead to the development of drugs that specifically target these proteins, reducing their harmful effects on brain cells and slowing the progression of Parkinson’s disease.
Additionally, the study provides a framework for exploring similar mechanisms in other neurodegenerative diseases, such as Alzheimer’s and Huntington’s. By identifying common pathways of cellular damage, scientists can work towards universal treatments that address multiple conditions. The ability to visualize these processes in real-time also offers a powerful tool for testing the efficacy of potential treatments, allowing for rapid adjustments and improvements in therapeutic approaches.
Cell Membrane’s Role in RAS Protein Interactions
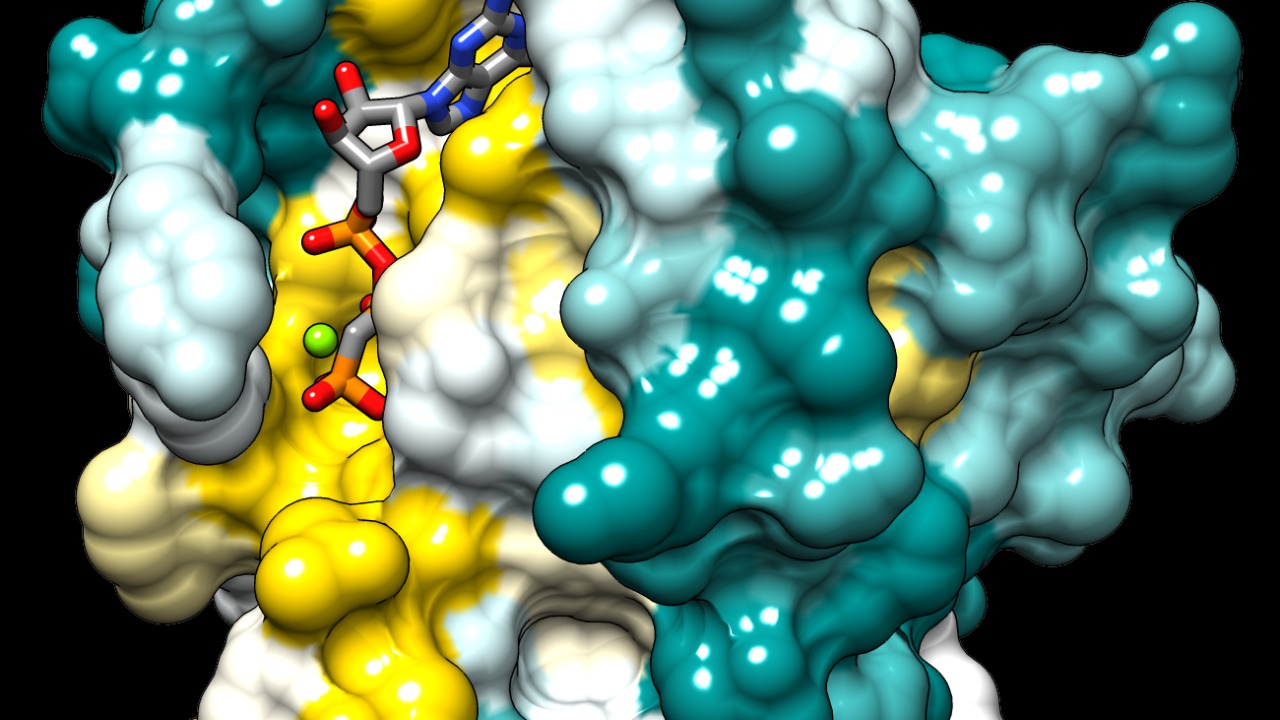
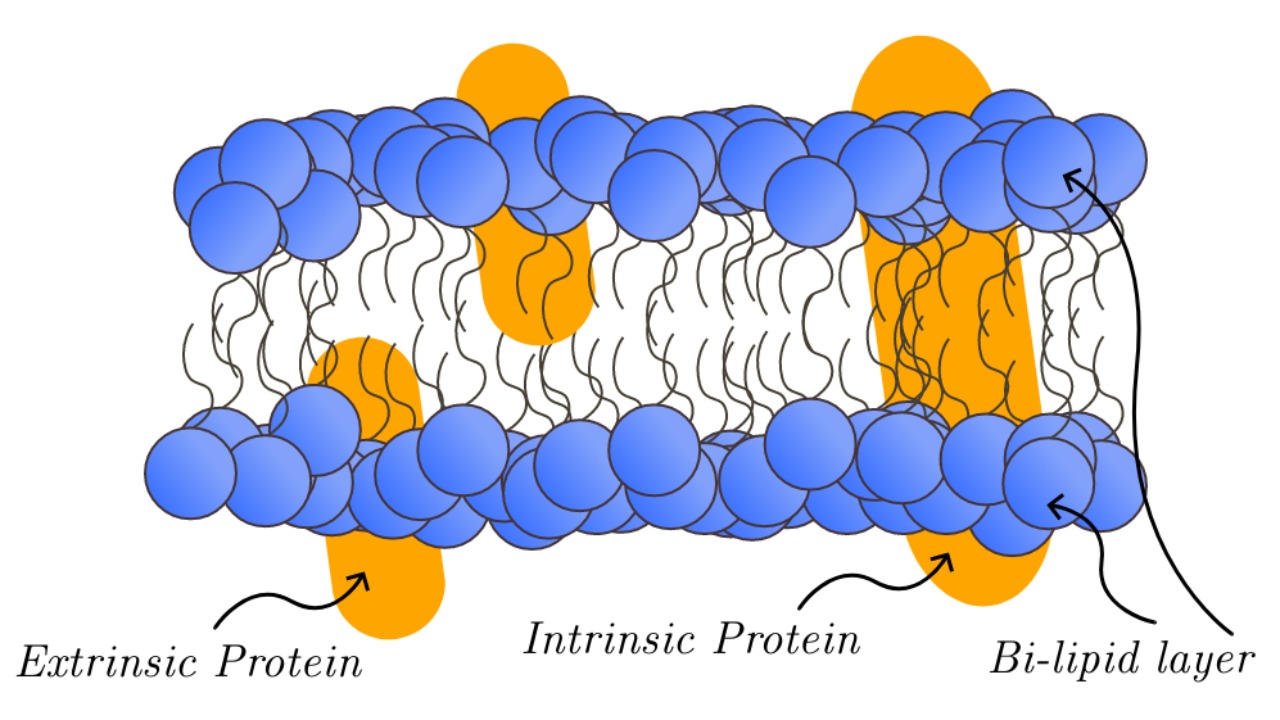
The cell membrane plays a crucial role in facilitating RAS protein interactions, as outlined in a study from May 18, 2020. This research emphasizes the importance of membrane location in protein signaling, demonstrating how spatial positioning enables specific protein behaviors (Salk Institute).
Examples from the study illustrate how membrane positioning affects RAS protein behavior, showing that these spatial dependencies are critical for proper cellular function. The “location, location, location” framework provides a valuable perspective on how membrane dynamics influence protein activation patterns, offering insights that are relevant to modern protein dynamics.
The study from the Salk Institute further elaborates on how the lipid composition of the cell membrane can influence RAS protein interactions. Variations in lipid types can alter the membrane’s physical properties, affecting how proteins like RAS are anchored and interact with other cellular components. This understanding is crucial for developing targeted therapies, as manipulating lipid environments could enhance or inhibit specific protein interactions, offering a novel approach to treating diseases driven by RAS signaling, such as certain cancers.
Moreover, the research highlights the potential for using synthetic biology to engineer cell membranes with tailored properties. By designing membranes that favor beneficial protein interactions, scientists could create cells with enhanced resistance to disease or improved therapeutic capabilities. This approach represents a significant step forward in precision medicine, where treatments are customized based on the unique cellular environments of individual patients.
Intersections with Neurodegenerative Research
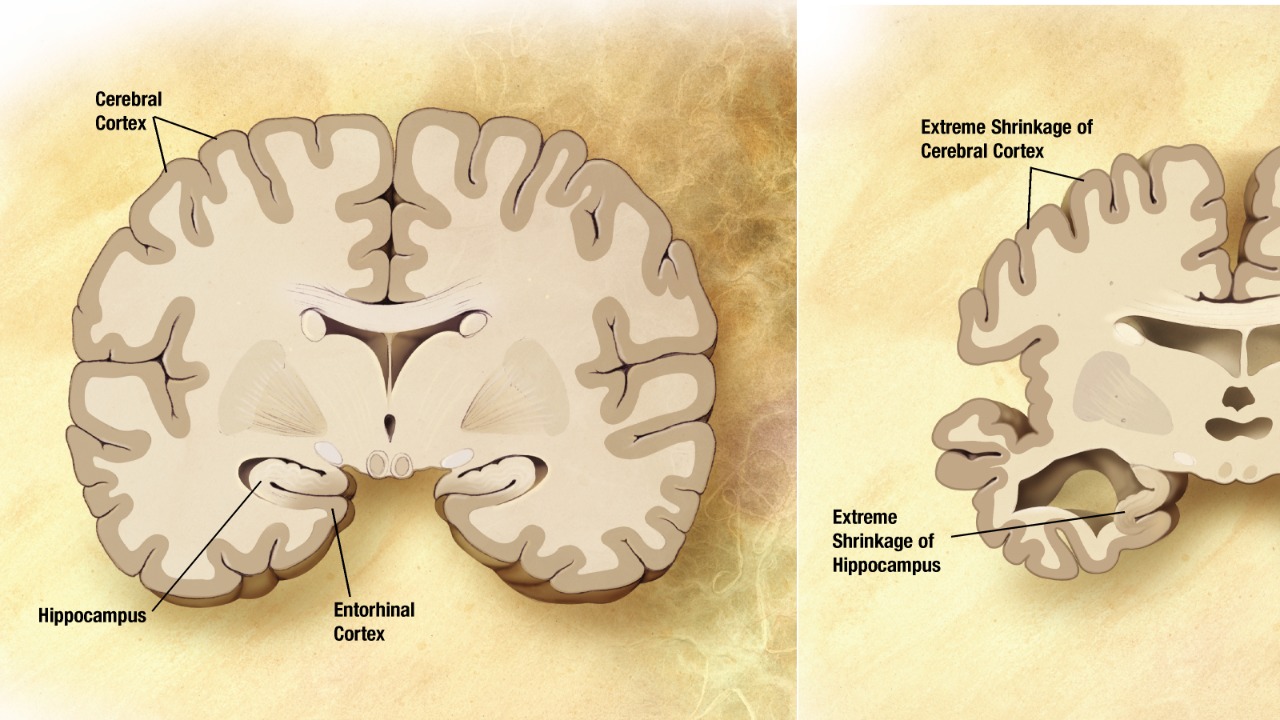
The convergence of findings from Parkinson’s protein studies and membrane protein dynamics suggests a unified model for understanding cellular vulnerability in neurodegenerative diseases. By examining how proteins interact with cell membranes, researchers can identify common factors that contribute to cellular damage across different conditions. This holistic view enables the development of broad-spectrum therapies that target these shared mechanisms, potentially offering relief for multiple neurodegenerative disorders.
Furthermore, the integration of these research areas highlights the importance of membrane integrity in maintaining cellular health. Disruptions in membrane structure or function can lead to a cascade of detrimental effects, emphasizing the need for therapies that preserve or restore membrane stability. As scientists continue to explore these intersections, they are likely to uncover new therapeutic targets and strategies that could revolutionize the treatment of neurodegenerative diseases.