For one of the most lethal brain cancers known, a simple nasal drop is emerging as an unexpectedly sophisticated weapon. Instead of relying on scalpels or high-dose chemotherapy, researchers are testing a therapy that slips in through the nose, travels along facial nerves, and attacks tumors from inside the brain’s own wiring.
The early data come from mice, not people, but the concept is striking: a noninvasive treatment that bypasses the blood-brain barrier and shrinks aggressive tumors that usually resist everything doctors throw at them. If the approach holds up in human trials, it could reshape how I think about treating brain cancer in the clinic.
Why glioblastoma is so hard to treat
Glioblastoma sits at the brutal intersection of biology and geography. It grows fast, infiltrates healthy brain tissue, and tends to recur even after surgeons remove every visible trace. Standard care usually combines surgery, radiation, and chemotherapy, yet survival is often measured in months rather than years, and the disease remains one of the most common and deadly primary brain tumors in adults, a reality underscored in recent coverage of nasal-drop research.
Part of the problem is access. Many drugs that work elsewhere in the body never reach effective levels in the brain because of the blood-brain barrier, a tightly regulated shield that keeps toxins out but also blocks helpful therapies. Even when oncologists push doses high enough to penetrate, patients pay the price in systemic side effects, while microscopic tumor cells hide in delicate brain regions that surgeons cannot safely touch. That combination of biological resistance and physical inaccessibility is what makes glioblastoma such a stubborn killer.
The surprising route: from nostrils to brain tumors
The new nasal-drop strategy tries to turn that anatomical disadvantage into an opportunity. Instead of forcing drugs through the bloodstream, scientists are delivering them directly into the nasal cavity, then letting them ride along the trigeminal and olfactory nerves that connect the face to the brain. In preclinical work from Washington University in St. Louis, investigators report that this intranasal route allowed therapeutic agents to reach deep-seated tumors in mice without surgery, a concept detailed in their description of noninvasive brain-tumor treatment.
What makes this pathway so intriguing is that it bypasses the blood-brain barrier entirely. Instead of squeezing molecules through tight junctions in blood vessels, the drops appear to move along nerve fibers and surrounding channels, effectively co-opting the brain’s own wiring as a delivery network. Researchers describe how this approach allowed them to concentrate therapy inside the tumor while limiting exposure to the rest of the body, a pattern echoed in institutional summaries of the same work from cancer specialists who highlight the potential of nasal delivery into brain tumors.
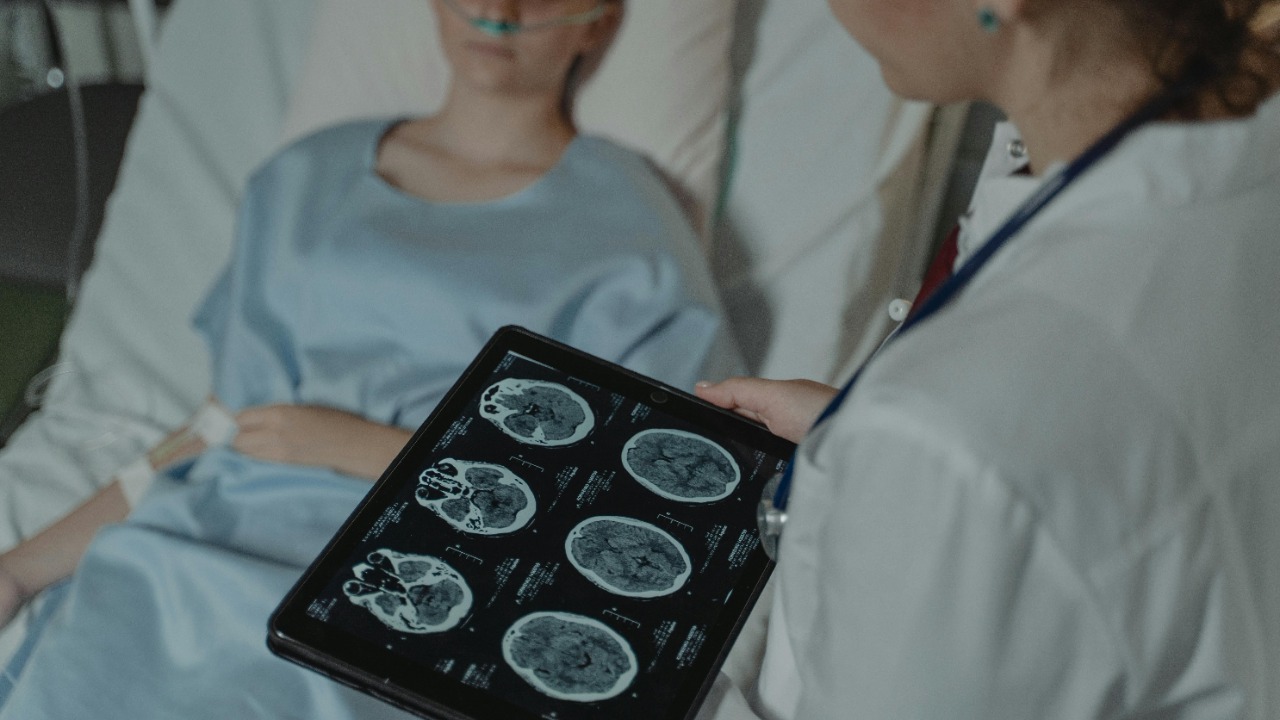
What the mouse experiments actually showed
In the mouse studies, the team focused on animals implanted with glioblastoma, a model chosen precisely because it mimics the rapid growth and invasiveness seen in human patients. After administering the nasal drops, they tracked how the therapy spread through the brain and monitored tumor size and survival. According to detailed research summaries, the treatment significantly reduced tumor burden in these mice and extended their lives compared with untreated controls, results that are laid out in reports on how nasal drops can fight brain tumors in mice.
The mechanism appears to rely on more than simple diffusion. Investigators describe the drops as effectively “hijacking” facial nerves to reach the tumor, then triggering immune responses or direct tumor cell death once they arrive. A separate explainer notes that the therapy co-opts these neural pathways to clear tumors in mouse models, reinforcing the idea that the nose-to-brain route is not just a shortcut but a targeted delivery system, as outlined in a broader discussion of how nasal drops can co-opt facial nerves for treatment.
How this approach differs from current brain cancer care
From a clinical perspective, the contrast with current glioblastoma care is stark. Today, patients typically undergo a craniotomy, weeks of radiation, and systemic chemotherapy that can cause fatigue, nausea, and blood count suppression, all while the tumor often finds ways to grow back. The nasal-drop strategy, by comparison, is designed to be noninvasive and repeatable, more like using an eye dropper than an operating room, a shift that institutional cancer centers have emphasized when describing these noninvasive nasal therapies.
Because the drops are delivered locally and travel along nerves, they may allow oncologists to use potent agents at the tumor site without flooding the rest of the body. That could open the door to combinations that would be intolerable if given intravenously, or to maintenance dosing that keeps microscopic disease in check after surgery and radiation. It is a fundamentally different philosophy of care, one that treats the nose as a controlled gateway into the brain rather than a barrier to be bypassed with ever more aggressive systemic drugs.
Why the findings are exciting, but still early
For all the enthusiasm, I have to keep the caveats front and center. These results come from mice, and history is full of cancer therapies that looked transformative in rodents but faltered in people. Mouse glioblastoma models, while useful, cannot fully capture the genetic diversity, immune environment, and treatment history of human patients who have already undergone surgery and radiation. Social media posts that highlight the work, including a physician’s summary of the nasal-drop mouse data, have been careful to note that translation to the clinic is not guaranteed.
Even so, the consistency of the preclinical signal has drawn attention beyond academic circles. A widely shared description of the experiments on a public page notes that researchers tested a nasal-drop therapy in mice with glioblastoma, a fast-growing tumor, and reported meaningful tumor shrinkage and survival gains, reinforcing the sense that this is more than a marginal tweak to existing methods, as summarized in a post describing nasal-drop therapy in glioblastoma mice. The challenge now is to design human trials that can test safety and dosing without overpromising on outcomes.
From lab bench to potential bedside
Moving from mouse models to human treatment will require more than academic enthusiasm. Drug delivery systems must be standardized, dosing schedules optimized, and regulatory pathways navigated. That process is already starting to take shape, with translational efforts focused on turning the nerve-hijacking concept into a platform that can carry different drugs or biologics into the brain. One research-focused blog describes how the same nasal route could be adapted for multiple agents, underscoring the flexibility of the approach as scientists refine the nose-to-brain delivery concept for future trials.
Commercial players are also beginning to circle the space. A clinical-trial news brief highlights how Neonc Technologies is developing a nasal-brain delivery system specifically for glioblastoma treatment, signaling that companies see enough promise to invest in hardware and formulations tailored to this route, as described in coverage of the firm’s nasal-brain delivery system. If those efforts align with academic protocols, patients could eventually see trials that combine cutting-edge delivery devices with therapies designed from the ground up for intranasal use.
What comes next for patients and clinicians
For patients and clinicians watching from the sidelines, the near-term impact is more about hope and direction than immediate change in standard care. Neurosurgeons, neuro-oncologists, and radiation specialists are still bound by existing guidelines, but many are already sharing the research within professional networks as an example of how creative delivery routes might finally crack the blood-brain barrier problem. One senior clinician amplified the findings on a professional networking site, framing the work as a notable advance in noninvasive brain-tumor therapy, a sentiment captured in a post about nasal drops fighting brain tumors.
As early-phase human studies emerge, I expect the key questions to center on safety, dosing frequency, and how well the therapy integrates with surgery, radiation, and systemic drugs rather than replacing them outright. If the nasal route proves reliable, it could become a new layer in multimodal care, perhaps used between MRI scans to keep residual disease in check or to deliver immunotherapies that need to sit directly in the tumor microenvironment. For now, the most honest assessment is that nasal drops are not a cure, but they are a credible, mechanistically grounded idea that could finally give doctors a way to reach one of cancer’s most inaccessible strongholds.
More from MorningOverview